As we have already seen nuclear power technology simply transforms stored energy in the nucleus of the atom to other forms of energy, such as heat. Because the energy liberated in a nuclear decay is many times more than any energy liberated in physical or chemical processes, this technology can offer a great deal more energy. Another very important aspect of this technology is that it can deliver energy either over a long time (as in the case of nuclear power reactors), or in fractions of a second (as in the case of nuclear explosions).
Clearly the best known use of nuclear technology in the military is in the use of nuclear weapons - from atomic and hydrogen bombs to ballistic missiles to neutron bombs. But, as in nuclear power plants, nuclear technology has been used in a number of military vehicles purely for power generation purposes. Of the non-explosive uses, the most widely used is nuclear power generators in nuclear submarines. Here, as in a electric power plant, the nuclear energy creates heat that is used for driving motors that propel the craft and generate its electricity. The relatively negligible fuel mass makes it ideal for use in crafts that remain in the water for long periods of time and do not require regular refueling. Below we will examine the use of nuclear energy for bomb making.
Details of Uranium Fission Reaction
Consider the uranium 235 fission reaction (see details to the right). Fission is initiated by the absorption of a slow moving neutron, which creates an excited uranium 236 nucleus. This nucleus is unstable, because similar to a growing liquid drop when a nucleus gets too large its outer nuclei have fewer neighboring ones to keep them whole nucleus in one large mass. (From the point of view of the binding energy, their binding energy is lower than what it would have been were the nucleons had been part of a smaller nucleus - remember that the binding energy per nucleon drops for these large nuclei.) In a too large a liquid drop the breakup results in formation of smaller droplets and not just the departure of separate surface molecules. Similarly, in nuclear fission the excited uranium 236 breaks into smaller nuclei; typically a krypton nucleus, a barium nucleus and three neutrons. The combined mass of these "parts" is less than the mass of the mother nucleus. This missing mass is released in the form of kinetic energy of the decay elements (the krypton, barium, and the three neutrons).
The above reaction is not the only possibility, however. Again, as in the liquid drop analogy, the uranium 236 could instead break into a xenon, a strontium, and two neutrons. Which of these two separate reactions will be the fate of a given uranium 236 nucleus could only be determined statistically. So, the energy and the number of the neutrons is statistical. Despite this statistical variation in a typical fission reaction about roughly 200 MeV of energy is released per single fission decay. This energy is distributed, roughly, in the following manner:
The role of any device that is designed for using fission energy is to extract the kinetic energy of the by products and to slow down the neutrons so that they in turn initiate further fission reactions and thus sustain a chain reaction. As the fission fragments collide with other surrounding nuclei they exchange kinetic energy and thus generate heat. In the case of power generating devices a heat exchange material, such as water, can carry away this heat and use it to generate steam. The steam can then mechanically turn a motor to generate electricity or to just turn a propeller. In the case of a bomb this energy is practically let loose to do damage. As for the neutrons, they can be slowed to velocities that could result in capture by other uranium 236 nuclei by making them collide with other nuclei first. These are called moderators. An ideal moderator must slow down the neutrons without absorbing them. Hydrogen is an excellent choice for slowing down the neutrons, but it is also a very good absorber! So, it is not a good moderator. Deuterium, on the other hand satisfies both criteria. So, one of the moderators used in reactors is heavy water. In heavy water the two hydrogen atoms of water molecule are in instead deuterium atoms. Other moderators are beryllium (Be) and graphite (carbon, 12C). As we have already discussed Control rods, often made of cadmium that is an effective neutron absorber, are brought near the fission material to affect the rate of the chain reaction, and thus control the rate of extracting energy form the uranium fuel.
Isotope enrichment/separation techniques
Uranium is a naturally occurring fissionable element. In its pure state it is shiny like a metal. It is very reactive and, like aluminum, quickly oxidizes. After mining it is chemically refined using acids to dissolve impurities. Uranium has 14 different isotopes, all of which are radioactive. But in its natural form on earth it typically has the following isotopic mixture, all of which are alpha emitters (so not very dangerous):
Only 235U isotope is capable of sustainable fission reactions, however. So, isotope enrichment methods have to be used to produce fissionable fuel. The exact percentage of the isotopes depends on the way that the fuel is to be used. Reactor grade uranium has a different mix of isotopes than does the weapon grade fuel.
One of the earliest methods used for the isotope separation was the electromagnetic method in a calutron mass spectrometer. In this device an electric field ionizes and accelerates the uranium atoms. (The uranium is first reacted with fluorine to produce uranium hexafluoride, UF6). This beam of ions are then introduced in a region of strong magnetic field that bends the path of charged particles. The more massive the ion, the less will its path bend. In this way unwanted isotopes can get separated to create a new sample with high density of the desired isotope as compared to the original natural sample. This is an easy method, but not very efficient. Other less costly methods have been devised and are currently in use.
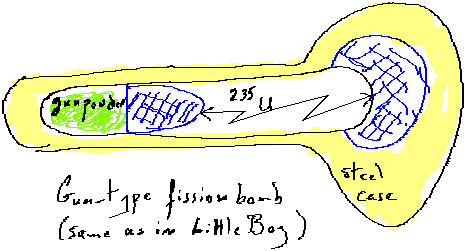
In the case of a power generating reactor the primary concern is to control the rate of fission and therefore the rate of heat generation from the reaction. Too much reaction could generate too much heat and melt the protection layers that separate the radioactive fuel from the environment. In the case of a bomb the concerns are very different. Part of this is because the aim of the bomb is to release as much energy as possible in as short a possible time and the other part is that there are no controllers to adjust the reaction rate. Because of these reasons the fuel must be in such a form that fission cannot occur (sub critical) until the time that the bomb is to explode (supercritical). Also, once the fuel is supercritical the fission has to occur as rapidly as possible, i.e. all possible fissionable nuclei should undergo fission.
The first of these goals is reached by controlling the density of the fuel. Through implosion the fuel density can be changed from a sub critical to a supercritical stage. Then neutron generating schemes can result in rapid fission process. Since collisions of alpha particles on beryllium can result in emission of slow neutrons, polonium, an alpha emitter, along with beryllium are introduced to the fuel at the stage when it reaches a supercritical stage. It is important to note that because of the issue of "critical mass" fission bombs cannot be made indefinitely large. Unfortunately, there is no such restriction on the size of fusion bombs.
Fission bombs are referred to as atomic bombs. Atomic processes deal mostly with the interaction of electrons with each other and with the nucleus through electromagnetic interactions. So fission bombs are not atomic, but in fact, nuclear bombs. The hydrogen bomb (H-bomb), similarly, is a nuclear device. Instead of fission, however, H-bombs are based on fusion reactions. There are other types of bombs that mix both of these processes. All of these highly destructive devices are based on nuclear reactions of fission and/or fusion.
In an H-bomb fission reaction triggers fusion of deuterium fuel (in the form of lithium deuteride), mostly through the radiation heat of the fission (hence the name: thermonuclear bomb). The energy of this bomb is primarily due to the deuterium-tritium fusion, but it also uses the fast moving neutrons generated from the fusion reaction to create another fission in the surrounding uranium 238 elements of the bomb.
For those of you who would like to learn more about the technical issues of nuclear weapons, I highly recommend the site set up by the Federation of American Scientists at: http://www.fas.org/nuke/hew.
Last Modified Monday, October 22, 2007 malekis@union.edu
n + 235 92U --------->236 92U* --------> 144 56Ba + 89 36Kr + 3 n
both Ba and Kr undergo a series of beta decays, as follows:
144 56Ba -------->144 57La ------->144 58Ce------->144 59Pr-------->144 60Nd
89 36Kr -------->89 37Rb --------> 89 38Sr --------> 89 39Y
In all of the above the superscript gives the nucleon (mass) number, A, and the subscript indicates the proton (charge) number, Z. The actual beta decays are of the type that results in increasing the proton number, i.e.: n (in parent nucleus) ----------> p (in daughter) + b- + ne , without changing the mass number.
For an animation of this reaction check out the appelet: Nuclear Decay Animation!